 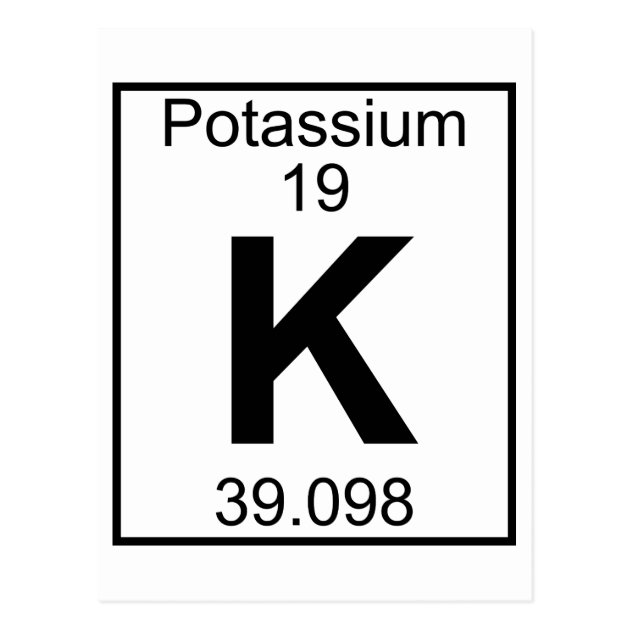
Cartoon by Nick D Kim (, used by permission). It has atomic number 19, element symbol K, and an atomic weight of 39.0983. The chemistry of potassium is dominated by electron loss to form K +. Potassium is an alkali metal, like sodium, cesium, and the other elements in group 1.It is the 7th most abundant element in the Earths crust. The picture above shows the colour arising from a burning mixture of potassium chlorate (KClO 3) and sucrose (only to be demonstrated by a professionally qualified chemist). In order to write the Potassium electron configuration we first need to know the number of electrons for the K atom (there are 19 electrons). Potassium and its salts impart a lilac colour to flames. It usually catches fire during the reaction with water. As do all the other metals of the alkali group, it decomposes in water with the evolution of hydrogen. It oxidises very rapidly in air and must be stored under argon or under a suitable mineral oil. It is silvery in appearance immediately after a fresh surface is exposed. It is one of the most reactive and electropositive of metals and, apart from lithium, it is the least dense known metal. Potassium is never found free in nature, but is obtained by electrolysis of the chloride or hydroxide, much in the same manner as prepared by Davy. 
Potassium is not found as a free element, so alchemists used it in the form of potash, which is potassium carbonate. It is also a vital element in the human diet. The alchemy symbol for potassium typically features a rectangle or open box ('goalpost' shape). Potassium is an essential constituent for plant growth and it is found in most soils. Potassiums symbol, K, comes from kalium, the Latin word for potash. Potassium is a metal and is the seventh most abundant and makes up about 1.5 % by weight of the earth's crust. The symbol of an element represents one atom of that element. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
